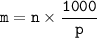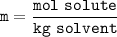Question 1 of 10
What would you need to do to calculate the molality of 10 mol of NaCl in 200
...

Question 1 of 10
What would you need to do to calculate the molality of 10 mol of NaCl in 200
mol of water?
A. Convert the 200 mol of water to kilograms of water.
B. Convert the 200 mol of water to liters of water.
C. Convert the 10 mol of NaCl to grams of NaCl.
O D. Convert the 10 mol of NaCl to kilograms of NaCl.
SUBMIT

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Which statements are true about electrolysis? check all that apply. electrolysis requires an acid be present. electrolysis is described by two half-reactions. electrolysis is not an industrial process. electrolysis results in commercially valuable products. electrolysis involves the transfer of electrons. reduction results in the loss of electrons. oxidation results in the loss of electrons.
Answers: 1

Chemistry, 21.06.2019 22:30
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 22.06.2019 02:40
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3

Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1
You know the right answer?
Questions

Chemistry, 07.04.2021 02:10




Mathematics, 07.04.2021 02:10

Mathematics, 07.04.2021 02:10


Mathematics, 07.04.2021 02:10




Mathematics, 07.04.2021 02:10

Mathematics, 07.04.2021 02:10


English, 07.04.2021 02:10

Biology, 07.04.2021 02:10