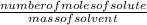Question 1 of 10
What would you need to do to calculate the molality of 10 mol of NaCl in 200
...

Question 1 of 10
What would you need to do to calculate the molality of 10 mol of NaCl in 200
mol of water?
A. Convert the 200 mol of water to kilograms of water.
B. Convert the 200 mol of water to liters of water.
C. Convert the 10 mol of NaCl to grams of NaCl.
O D. Convert the 10 mol of NaCl to kilograms of NaCl.
SUBMIT

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:30
Apump contains 0.5 l of air at 203 kpa.you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2

Chemistry, 22.06.2019 11:00
The number to the right of an element's symbol (ex. c-12) identifies the of an isotope.
Answers: 1


Chemistry, 22.06.2019 19:10
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2
You know the right answer?
Questions





English, 04.01.2021 02:20


Mathematics, 04.01.2021 02:20

Business, 04.01.2021 02:20


Computers and Technology, 04.01.2021 02:20






Computers and Technology, 04.01.2021 02:20


Mathematics, 04.01.2021 02:20