
Chemistry, 14.11.2020 03:10 gjaime1307
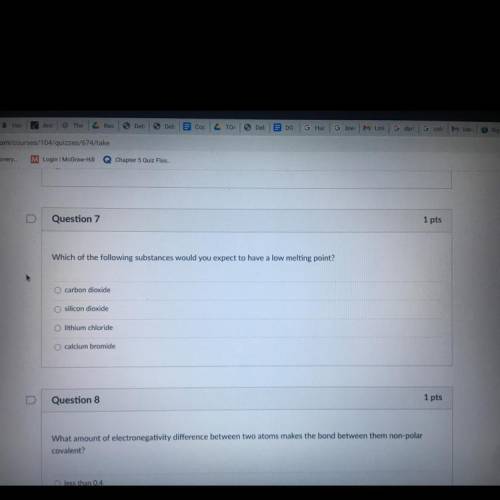
Which of the following substances would you expect to have a low melting point?
A. carbon dioxide
B. silicon dioxide
C. lithium chloride
D. calcium bromide

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1

Chemistry, 22.06.2019 19:00
Sum of brother and sisters age is 26. four times the brothers age is subtracted from three times the sisters age, the difference is 8. what are the ages of the brother and sister?
Answers: 1

Chemistry, 22.06.2019 19:00
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3
You know the right answer?
Which of the following substances would you expect to have a low melting point?
A. carbon dioxide
Questions

Mathematics, 12.12.2020 16:50

Computers and Technology, 12.12.2020 16:50

History, 12.12.2020 16:50

Mathematics, 12.12.2020 16:50

Mathematics, 12.12.2020 16:50



English, 12.12.2020 16:50

Mathematics, 12.12.2020 16:50

Engineering, 12.12.2020 16:50

Health, 12.12.2020 16:50




Mathematics, 12.12.2020 16:50



Mathematics, 12.12.2020 16:50

Biology, 12.12.2020 16:50

Mathematics, 12.12.2020 16:50



