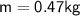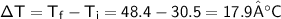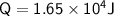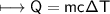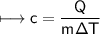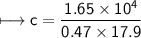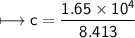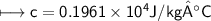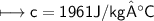Chemistry, 13.11.2020 09:10 naeaamm2528
A chemist carefully measures the amount of heat needed to raise the temperature of a 0.47 kg sample of C6H7N from 30.5 degrees C to 48.4 degrees C. The experiment shows that 1.65 x 10^4 J of heat are needed. What can the chemist report for the molar heat capacity of C6H7N? Be sure your answer has the correct number of significant digits.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 20:20
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1

Chemistry, 22.06.2019 21:00
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3

Chemistry, 22.06.2019 21:30
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3
You know the right answer?
A chemist carefully measures the amount of heat needed to raise the temperature of a 0.47 kg sample...
Questions



Arts, 02.03.2021 23:00



English, 02.03.2021 23:00

English, 02.03.2021 23:00


Chemistry, 02.03.2021 23:00

History, 02.03.2021 23:00

History, 02.03.2021 23:00



Mathematics, 02.03.2021 23:00


English, 02.03.2021 23:00