
Chemistry, 12.11.2020 06:20 Lavibookman7934
Using the equations
2 Fe (s) + 3 Cl₂ (g) → 2 FeCl₃ (s) ∆H° = -800.0 kJ/mol
Si(s) + 2 Cl₂ (g) → SiCl₄ (s) ∆H° = -640.1 kJ/mol
Determine the enthalpy (in kJ/mol) for the reaction
3 SiCl₄ (s) + 4 Fe (s) → 4 FeCl₃ (s) + 3 Si (s)

Answers: 3


Another question on Chemistry




You know the right answer?
Using the equations
2 Fe (s) + 3 Cl₂ (g) → 2 FeCl₃ (s) ∆H° = -800.0 kJ/mol
Si(s) + 2 Cl₂ (g)...
Si(s) + 2 Cl₂ (g)...
Questions

Chemistry, 15.10.2019 06:20

History, 15.10.2019 06:20


Mathematics, 15.10.2019 06:20

Mathematics, 15.10.2019 06:20

Chemistry, 15.10.2019 06:30

Health, 15.10.2019 06:30



Mathematics, 15.10.2019 06:30

Physics, 15.10.2019 06:30


Biology, 15.10.2019 06:30


English, 15.10.2019 06:30




Mathematics, 15.10.2019 06:30

Mathematics, 15.10.2019 06:30

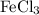 has been 320.3 kJ/mol.
has been 320.3 kJ/mol.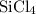 , and 4 moles of Fe.
, and 4 moles of Fe. 640.1 kJ/mol.
640.1 kJ/mol.

