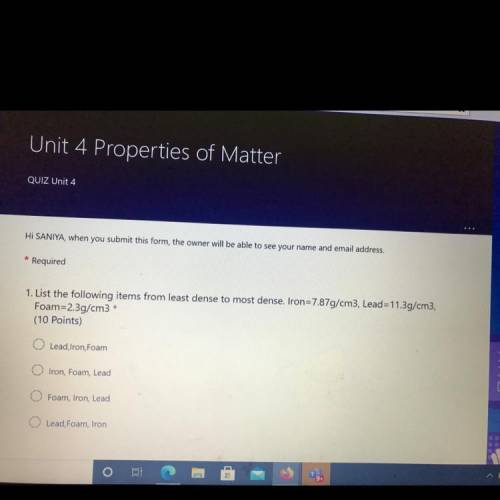
:answer question number 1
...

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
200. ml of 3.00 m nacl solution is diluted to a final volume of 500. ml. what is the molarity of the final solution?
Answers: 2

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 04:00
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 11:40
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2
You know the right answer?
Questions

Chemistry, 30.01.2021 03:40

Arts, 30.01.2021 03:40




Mathematics, 30.01.2021 03:40

Advanced Placement (AP), 30.01.2021 03:40

Arts, 30.01.2021 03:40





Mathematics, 30.01.2021 03:40


Physics, 30.01.2021 03:40


Mathematics, 30.01.2021 03:40


Mathematics, 30.01.2021 03:40

Biology, 30.01.2021 03:40