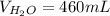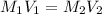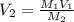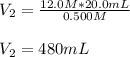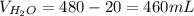Chemistry, 10.11.2020 17:00 victoria6929
Determine the volume of distilled water that should be added to 20.0 mL of 12.0 M HCl(aq) in order to prepare a 0.500 M HCl(aq) solution.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3


Chemistry, 23.06.2019 06:30
(04.01 lc) which of the following is true about science? (5 points) select one: a. it is not influenced by social conditions. b. it is not determined by external local factors. c. political conditions are unable to influence it. d. economic concerns may prevent it from solving problems.
Answers: 1

Chemistry, 23.06.2019 08:00
Can anyone answer these questions? ? i need it before 1: 00pm today
Answers: 3
You know the right answer?
Determine the volume of distilled water that should be added to 20.0 mL of 12.0 M HCl(aq) in order t...
Questions


Mathematics, 18.10.2020 08:01




Mathematics, 18.10.2020 08:01



History, 18.10.2020 08:01

English, 18.10.2020 08:01




Computers and Technology, 18.10.2020 08:01




Social Studies, 18.10.2020 08:01