Can someone please help me answer that question
...

Chemistry, 10.11.2020 01:00 jkjjoijjm5928
Can someone please help me answer that question
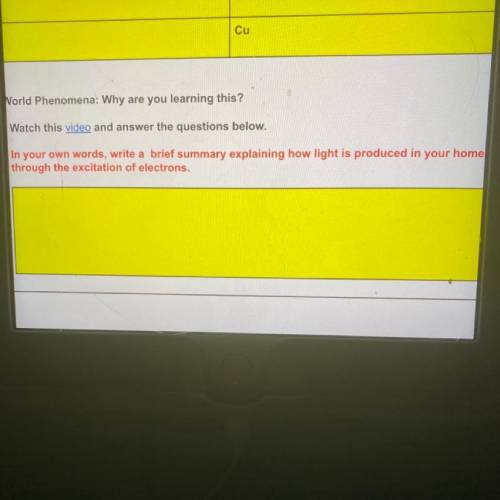

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2

You know the right answer?
Questions


Physics, 13.06.2021 16:00

Social Studies, 13.06.2021 16:00

Computers and Technology, 13.06.2021 16:00


Geography, 13.06.2021 16:00

History, 13.06.2021 16:00


English, 13.06.2021 16:00




Mathematics, 13.06.2021 16:00

Chemistry, 13.06.2021 16:00


Social Studies, 13.06.2021 16:00

Mathematics, 13.06.2021 16:10



Physics, 13.06.2021 16:10



