
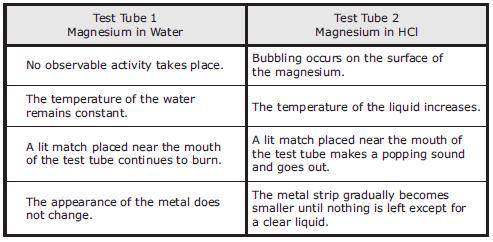
A student obtains two strips of magnesium, Mg, ribbon that are each 3 cm long. One strip of magnesium is placed in a test tube containing 5 mL of water, and the other strip is placed in a test tube containing 5 mL of hydrochloric acid, HCl. Both liquids are at room temperature. The student’s observations are recorded in the table.
Which statement is NOT supported by the student's observations?
Question 5 options:
A chemical reaction takes place between magnesium and hydrochloric acid.
A gas is released in Test Tube 2.
The substances in both test tubes are reactive only at high temperatures.
Energy is released in the reaction involving hydrochloric acid.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 07:00
How far is the region from the equator or control climate
Answers: 1

Chemistry, 22.06.2019 08:30
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3
You know the right answer?
A student obtains two strips of magnesium, Mg, ribbon that are each 3 cm long. One strip of magnesiu...
Questions


History, 28.07.2019 04:34


History, 28.07.2019 04:34





History, 28.07.2019 04:34

Mathematics, 28.07.2019 04:34





English, 28.07.2019 04:34

Chemistry, 28.07.2019 04:34




Spanish, 28.07.2019 04:34




