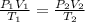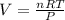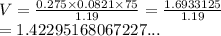Chemistry, 09.11.2020 06:10 dakotacsey03
If I have 0.275 moles of gas at a temperature of 75 K and a pressure of 17.5 psi, what is the volume of the gas?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:00
What postulate of the kinetic molecular theory best explains why gases have high fluidity? because collisions between gas particles are elastic, there is no loss of energy as particles flow past each other. because gases consist of large numbers of tiny particles, they spread out and do not come in contact with each other. because the attractive forces between gas particles are negligible, gas particles can glide easily past one another. because the average kinetic energy of gas particles increases as temperature increases, gas particles behave more like a liquid. question 6 compare the compressibility of gases and liquids. support your answer by describing the arrangement of particles in gases and liquids.
Answers: 1

Chemistry, 21.06.2019 17:40
In the reading, yao chen-yuan describes traveling to deliver a message. why was he willing to risk danger to travelto tientsin? he wanted to the boxers with their cause
Answers: 2

Chemistry, 21.06.2019 23:00
Layers of rock containing fossils, like the layers illustrated here, are most likely composed of rocks.
Answers: 2

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1
You know the right answer?
If I have 0.275 moles of gas at a temperature of 75 K and a pressure of 17.5 psi, what is the volume...
Questions




Spanish, 30.03.2020 18:44

Mathematics, 30.03.2020 18:44

Mathematics, 30.03.2020 18:44


Health, 30.03.2020 18:44


Social Studies, 30.03.2020 18:44


History, 30.03.2020 18:44


Mathematics, 30.03.2020 18:44

Chemistry, 30.03.2020 18:44



Mathematics, 30.03.2020 18:44


Mathematics, 30.03.2020 18:45