Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 05:30
A3.37-mg sample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0 ml. then 10.0 ml of this solution was placed in a 50-ml volumetric flask and treated with 5 ml of phenol solution plus 2 ml of sodium hypochlorite solution. the sample was diluted to 50.0 ml, and the absorbance at 625 nm was measured in a 1.00-cm cuvette and found to be 0.486. for reference, a standard solution was prepared from 10.0 mg of nh4cl (molar mass = 53.49 grams/mole) dissolved in 1.00 l of water. then 10.0 ml of this standard was placed in a 50-ml volumetric flask, treated in the same manner as the unknown, and the absorbance found to be 0.323. finally, a reagent blank was prepared using distilled water in place of unknown, it was treated in the same manner as the unknown, and the absorbance found to be 0.076. calculate the weight percent of nitrogen in the protein.
Answers: 1

Chemistry, 22.06.2019 06:00
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 23:00
What is the number of neutrons in an atom with atomic mass of 35
Answers: 2
You know the right answer?
WILL MARK BRAINLIEST:
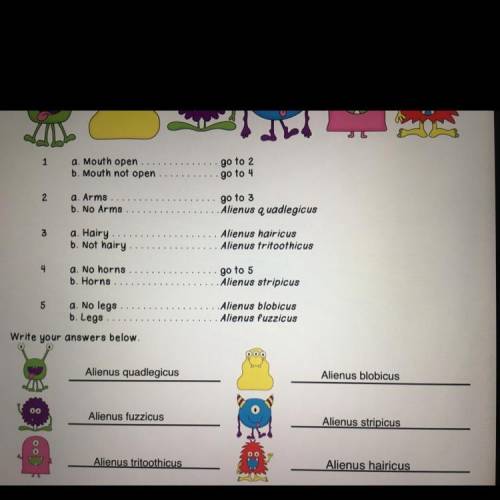
Identify the dichotomous key: plz ::: Each of these aliens belongs to the sam...
Questions

Mathematics, 08.11.2020 08:20


English, 08.11.2020 08:20

Computers and Technology, 08.11.2020 08:20




Mathematics, 08.11.2020 08:30


Mathematics, 08.11.2020 08:30


German, 08.11.2020 08:30

Health, 08.11.2020 08:30



Mathematics, 08.11.2020 08:30


Mathematics, 08.11.2020 08:30


English, 08.11.2020 08:30