
Chemistry, 06.11.2020 20:00 thibeauxkristy
A sample of 87.6 g of carbon is reacted with 136 g of
fluorine gas to produce carbon tetrafluoride. Using
the balanced equation below, predict which is the
limiting reactant and the maximum amount in moles
of carbon tetrafluoride that can be produced.
C +2F2 → CF4
A. fluorine, 1.79 moles
B. carbon, 7.29 moles
C. fluorine, 6.72 moles
D. carbon, 4.63 moles

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 10:00
Main expenses you plan on making payments on a new car too. you want to spend 15% of your monthly net pay on the car payment, insurance, registration, and taxes combined. what is your monthly car allowance? $149.46 $298.91 $448.37 $597.83
Answers: 3


Chemistry, 22.06.2019 17:30
Energy defines the different "states" of matter. in no more than 3 sentences, describe the amount of kinetic energy that each of the 3 states of matter possesses and relate that to the atom/molecular motion of each "state".
Answers: 2
You know the right answer?
A sample of 87.6 g of carbon is reacted with 136 g of
fluorine gas to produce carbon tetrafluoride....
Questions


Business, 16.01.2020 05:31

Physics, 16.01.2020 05:31

Mathematics, 16.01.2020 05:31


Mathematics, 16.01.2020 05:31


Mathematics, 16.01.2020 05:31

Mathematics, 16.01.2020 05:31

Mathematics, 16.01.2020 05:31


Mathematics, 16.01.2020 05:31

Mathematics, 16.01.2020 05:31



Biology, 16.01.2020 05:31

Mathematics, 16.01.2020 05:31

Mathematics, 16.01.2020 05:31

Spanish, 16.01.2020 05:31

History, 16.01.2020 05:31

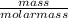
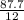 = 7.31moles
= 7.31moles 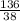 = 3.58moles
= 3.58moles 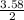 = 1.79moles of CF₄
= 1.79moles of CF₄

