
Chemistry, 06.11.2020 17:50 alexa006ox9k63
Determine the maximum amount of NaNO3 that was produced during the experiment. Explain how you determined this amount

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1

Chemistry, 22.06.2019 17:50
You exhale co2 which is produced during cellular respiration. co2 combines with the water in your blood's plasma to make up one half of the body's most important buffer pair, carbonic acid. the more physical activity you engage in, the more co2 your body is producing. you can see this by putting some of the cabbage indicator in a glass and then blowing bubbles into it through a straw. can you see a change in the color of the indicator?
Answers: 2

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1
You know the right answer?
Determine the maximum amount of NaNO3 that was produced during the experiment. Explain how you deter...
Questions

Mathematics, 05.10.2021 14:00

English, 05.10.2021 14:00


Biology, 05.10.2021 14:00

Mathematics, 05.10.2021 14:00

Social Studies, 05.10.2021 14:00


English, 05.10.2021 14:00

Mathematics, 05.10.2021 14:00

Mathematics, 05.10.2021 14:00







Mathematics, 05.10.2021 14:00


Physics, 05.10.2021 14:00

Mathematics, 05.10.2021 14:00

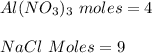
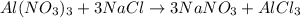
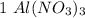 mole reacts in
mole reacts in 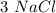 moles to give
moles to give 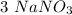 moles and
moles and 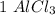 mole.
mole. 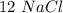 moles,
moles, 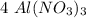 moles react totally. And though we got
moles react totally. And though we got 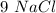 moles.
moles. 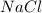 is the only reagent.
is the only reagent. 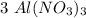 moles to give
moles to give 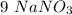 moles and
moles and 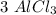 moles.
moles. 


