
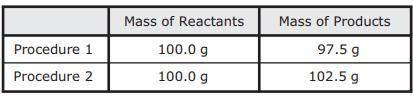
As part of an investigation, students combined substances in a beaker to observe chemical reactions. They performed two procedures. They measured the mass of each substance before and after each reaction. The table shows their observations.
Assuming the students did not make any careless errors, what likely explains these changes in mass?
A. Procedure 1: All the reactants were liquids that evaporated.
Procedure 2: A gas was formed as one product, and it escaped into the air.
B. Procedure 1: One of the reactants was converted to thermal energy.
Procedure 2: All the products were liquids.
C. Procedure 1: The reactants were liquids with different densities.
Procedure 2: The reactants were combined into only one product.
D. Procedure 1: One of the products was a gas that escaped into the air.
Procedure 2: A gas from the air reacted with one of the other reactants.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 09:40
Apiece of copper has a temperature of 75.6 0c. when the metal is placed in 100.0 grams of water at 19.1 0c, the temperature rises by 5.5 0c. what is the mass of the metal?
Answers: 1

Chemistry, 22.06.2019 15:30
Which of the following are correct values for the ideal gas laws constant r
Answers: 1

Chemistry, 22.06.2019 17:00
The msds for glacial acetic acid says that it is a flammable liquid that can severely burn any human tissue it comes in contact with. it reacts with bases, various metals, and strong oxidizing agents. its vapors can form explosive mixtures with air.
Answers: 1
You know the right answer?
As part of an investigation, students combined substances in a beaker to observe chemical reactions....
Questions






English, 31.01.2020 20:55

Mathematics, 31.01.2020 20:55

History, 31.01.2020 20:55





Mathematics, 31.01.2020 20:55

Mathematics, 31.01.2020 20:55



History, 31.01.2020 20:55


Mathematics, 31.01.2020 20:55



