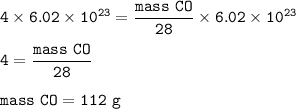The mass of CO = 112 g
Further explanation
The mole is the number of particles contained in a substance
1 mol = 6.02.10²³ particles
Moles can also be determined from the amount of substance mass and its molar mass
number of atoms in 88 g CO₂ :
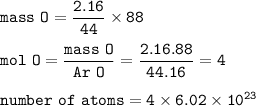
number of atoms in CO :
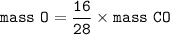
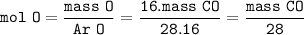
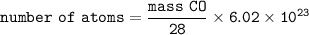
number of atoms in CO₂ = number of atoms in CO :