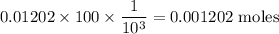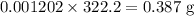Chemistry, 03.11.2020 16:30 elwinelwin9475
The final volume of buffer solution must be 100.00 mL and the final concentration of the weak acid must be 0.100 M. Based on this information, what mass of solid conjugate base should the student weigh out to make the buffer solution with a pH of 1.00

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
Given that the molar mass of nano3 is 85.00 g/mol, what mass of nano3 is needed to make 4.50 l of a 1.50 m nano3solution? use .6.75 g18.9 g255 g574 g
Answers: 1

Chemistry, 21.06.2019 21:00
Which of the following compounds does not contain molecules? question 2 options: co2 h2 nacl h2o
Answers: 1

Chemistry, 22.06.2019 04:00
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3

Chemistry, 23.06.2019 01:00
Which fossil fuel is mainly used for heating and cooking? a. electricity b. coal c. petroleum d. natural gas
Answers: 2
You know the right answer?
The final volume of buffer solution must be 100.00 mL and the final concentration of the weak acid m...
Questions


Mathematics, 16.02.2021 05:30

Biology, 16.02.2021 05:30


Mathematics, 16.02.2021 05:30

Mathematics, 16.02.2021 05:30

Mathematics, 16.02.2021 05:30







Mathematics, 16.02.2021 05:30


Mathematics, 16.02.2021 05:30

Social Studies, 16.02.2021 05:30


Mathematics, 16.02.2021 05:30

Mathematics, 16.02.2021 05:30

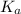 = Acid dissociation constant =
= Acid dissociation constant = 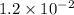
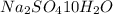 )
)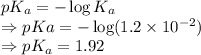
![pH=pK_a+\log\dfrac{[A^-]}{[HA]}\\\Rightarrow pH-pK_a=\log\dfrac{[A^-]}{[HA]}\\\Rightarrow 10^{pH-pK_a}=\dfrac{[A^-]}{[HA]}\\\Rightarrow [A^-]=10^{pH-pK_a}[HA]\\\Rightarrow [A^-]=10^{1-1.92}\times0.1\\\Rightarrow [A^-]=0.01202\ \text{M}](/tpl/images/0863/3238/f5459.png)