
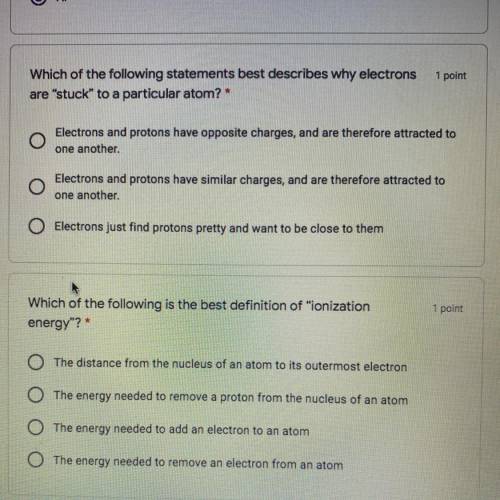
Which of the following statements best describes why electrons
are “stuck" to a particular atom? *
•Electrons and protons have opposite charges, and are therefore attracted to
one another.
Electrons and protons have similar charges, and are therefore attracted to
one another.
Electrons just find protons pretty and want to be close to them

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 10:00
A50.0g sample of liquid water at 0.0 c ends up as ice at -20.0 c. how much energy is involved in this change?
Answers: 1

Chemistry, 22.06.2019 15:20
Select the most likely product for this reaction: koh(aq) + co2(g) – ? k2co3(aq) + h2o(1) k(s) + h2(g) + o2(g) k(s) + co3(9) +h2
Answers: 2
You know the right answer?
Which of the following statements best describes why electrons
are “stuck" to a particular atom? *<...
Questions


Computers and Technology, 30.03.2020 18:57









Computers and Technology, 30.03.2020 18:58







Mathematics, 30.03.2020 18:58

Mathematics, 30.03.2020 18:58



