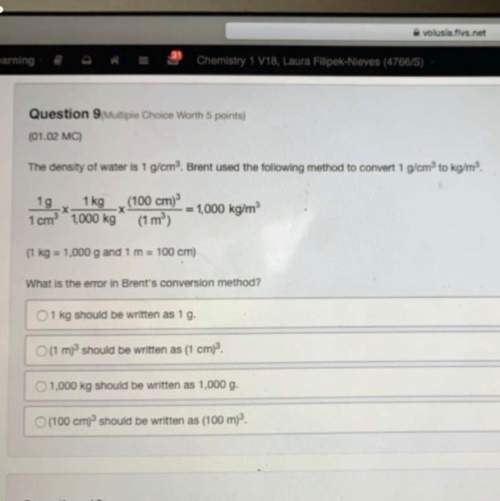
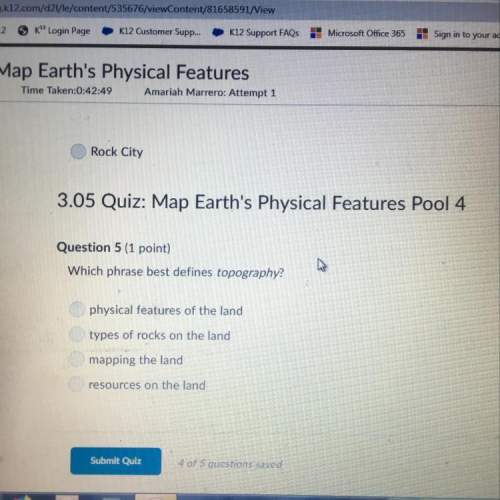
A chemist prepares a solution of aluminum sulfite by measuring out of aluminum sulfite into a volumetric flask and filling the flask to the mark with water. Calculate the concentration in of the chemist's aluminum sulfite solution. Round your answer to significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:50
Achemist has dissolved a certain substance in water. the chemist knows that more of the substance could be dissolved into the water before it stops dissolving. therefore
Answers: 2

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 11:10
Which of the following shapes would represent a molecule with two bonded atoms and 3 lone pairs on only one of them , trigonal planar , bent , trigonal pyramidal , linear
Answers: 1

Chemistry, 22.06.2019 14:20
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1
You know the right answer?
A chemist prepares a solution of aluminum sulfite by measuring out of aluminum sulfite into a volume...
Questions

Advanced Placement (AP), 29.01.2021 16:30


Chemistry, 29.01.2021 16:30

History, 29.01.2021 16:30

Mathematics, 29.01.2021 16:30

Physics, 29.01.2021 16:30



Mathematics, 29.01.2021 16:30



English, 29.01.2021 16:30




Mathematics, 29.01.2021 16:30

Biology, 29.01.2021 16:30

Mathematics, 29.01.2021 16:30

History, 29.01.2021 16:30