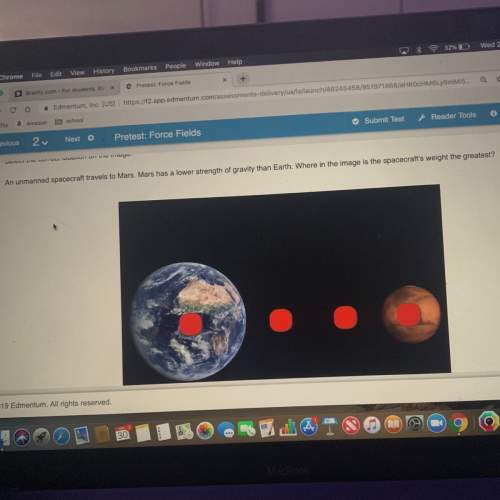
Which additional product balances the reaction H2SO4 + 2NaOH - Na2SO4 + ?
0 2H²0
O 20H
O...

Chemistry, 30.10.2020 21:40 naomicervero
Which additional product balances the reaction H2SO4 + 2NaOH - Na2SO4 + ?
0 2H²0
O 20H
O H₂O₂
O H₂O

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 22.06.2019 22:30
The vapor pressure of ethanol is 1.00 × 102 mmhg at 34.90°c. what is its vapor pressure at 61.61°c? (δhvap for ethanol is 39.3 kj/mol.)
Answers: 2

Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1
You know the right answer?
Questions


English, 12.08.2021 14:50

English, 12.08.2021 14:50

English, 12.08.2021 14:50

Mathematics, 12.08.2021 14:50


English, 12.08.2021 14:50

Computers and Technology, 12.08.2021 14:50



Mathematics, 12.08.2021 15:20



Mathematics, 12.08.2021 15:20

Computers and Technology, 12.08.2021 15:20





Engineering, 12.08.2021 15:20