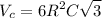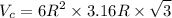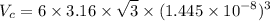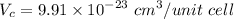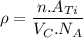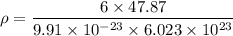Chemistry, 29.10.2020 17:10 estefaniapenalo
Titanium has an HCP unit cell for which the ratio of the lattice parameters c/a is 1.58. If the radius of the Ti atom is 0.1445 nm, calculate the density of Ti and compare it with the literature value of 4.51 g/cm3 . The atomic mass of titanium is 47.87 g/mol.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:10
What approach is required to balance the objectives of sustainable development? balancing the objectives of sustainable development requires a(n) .
Answers: 3

Chemistry, 22.06.2019 03:50
What is the temperature of one mole of helium gas at stp?
Answers: 3

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2
You know the right answer?
Titanium has an HCP unit cell for which the ratio of the lattice parameters c/a is 1.58. If the radi...
Questions

Mathematics, 31.10.2019 22:31

Mathematics, 31.10.2019 22:31




Mathematics, 31.10.2019 22:31


Mathematics, 31.10.2019 22:31

Mathematics, 31.10.2019 22:31






Mathematics, 31.10.2019 22:31


Mathematics, 31.10.2019 22:31

Mathematics, 31.10.2019 22:31

Mathematics, 31.10.2019 22:31

English, 31.10.2019 22:31