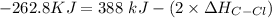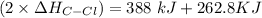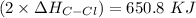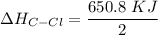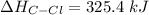Chemistry, 28.10.2020 17:10 monkemily1
Compute the bond energy of the C-Cl bond using the reaction and data in question 10, the value of the bond energy for the C-H bond of 410 kJ/mol and the value of the H-H bond of 432 kJ/mol. The C-Cl bond energy in kJ/mol is:

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:50
What is the temperature of one mole of helium gas at stp?
Answers: 3

Chemistry, 22.06.2019 05:30
Which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Answers: 2

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2
You know the right answer?
Compute the bond energy of the C-Cl bond using the reaction and data in question 10, the value of th...
Questions










Mathematics, 23.12.2021 05:00


English, 23.12.2021 05:00




Computers and Technology, 23.12.2021 05:00

SAT, 23.12.2021 05:00



Mathematics, 23.12.2021 05:10

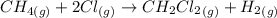
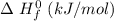
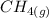 - 74.8
- 74.8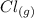 120.9
120.9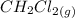 -95.8
-95.8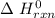 can be computed as follows:
can be computed as follows: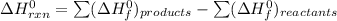
![\Delta H^0_{rxn } = [ 1 \ mol \times ( \Delta H^0_{CH_2Cl_{2(g)}}) + 1 \ mol \times ( \Delta H^0_{H_{2(g)}})] - [ 1 mol \times (\Delta H^0_{CH_4}_{(g)} +2 \ mol \times (\Delta \ H^0 _{Cl(g)})]}](/tpl/images/0847/6940/e9f6c.png)
![\Delta^0_{rxn } = [ 1 \ mol \times ( -95.8 \ kJ/mol) + 1 \ mol \times ( 0 \ kJ/mol] - [ 1 mol \times (-74.8 \ kJ/mol +2 \ mol \times (120.9 \ kJ/mol)]}](/tpl/images/0847/6940/36775.png)
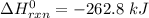
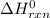 to be -262.8 kJ
to be -262.8 kJ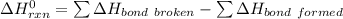
![-262. 8 KJ =[4 \times ( \Delta H_{CH})] - 1 [ (2 \times \Delta H_{CH} ) + ( 2 \times \Delta H_{C-Cl}) + (1 + \Delta H _{H-H})]](/tpl/images/0847/6940/9b723.png)
![-262. 8 KJ =[4 \times ( 410 \ kJ/mol) ] - 1 [ (2 \times 410 \ kJ/mol ) + ( 2 \times \Delta H_{C-Cl}) + (1 +432 \ kJ/mol]](/tpl/images/0847/6940/aee67.png)