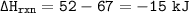Chemistry, 27.10.2020 14:00 snicklegirlp4isqr
In a certain chemical reaction, the bond energy of the products is 67 kJ and
that of the reactants is 52 kJ. Because energy is conserved, how much
potential energy does the system release or absorb?
O A. The reaction uses up 15 kJ of energy from the reaction system.
B. The reaction creates 15 kJ of energy when bonds form.
C. The reaction system absorbs 15 kJ of energy from the
surroundings.
O D. The surroundings absorb 15 kJ of potential energy.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
What does x represent in the formula for the compound xcl4?
Answers: 2

Chemistry, 22.06.2019 10:30
Apiece of metal with a length of 1.42 cm was measured using four different instruments. which of the following measurements is the most accurate?
Answers: 3


Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1
You know the right answer?
In a certain chemical reaction, the bond energy of the products is 67 kJ and
that of the reactants...
Questions


Biology, 26.08.2019 19:30

Chemistry, 26.08.2019 19:30



Mathematics, 26.08.2019 19:30

Mathematics, 26.08.2019 19:30


English, 26.08.2019 19:30

Spanish, 26.08.2019 19:30


Mathematics, 26.08.2019 19:30


Geography, 26.08.2019 19:30

Mathematics, 26.08.2019 19:30



Mathematics, 26.08.2019 19:30