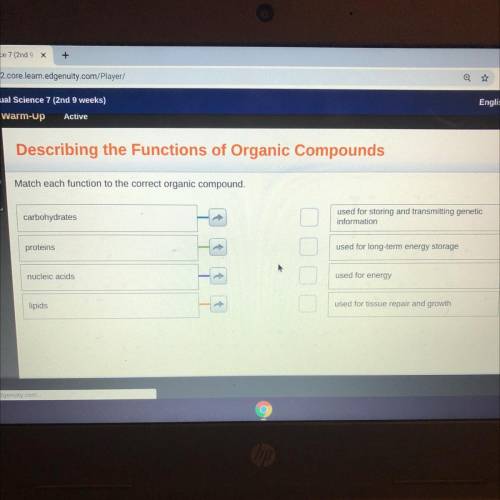
Plsss help me and give me the right answers... I will give brainless and 30 points.
...


Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 20:30
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2

You know the right answer?
Questions



Social Studies, 13.01.2021 17:50

Mathematics, 13.01.2021 17:50

English, 13.01.2021 17:50


Mathematics, 13.01.2021 17:50




Health, 13.01.2021 17:50

Social Studies, 13.01.2021 17:50