
Chemistry, 23.10.2020 15:20 BaileyElizabethRay
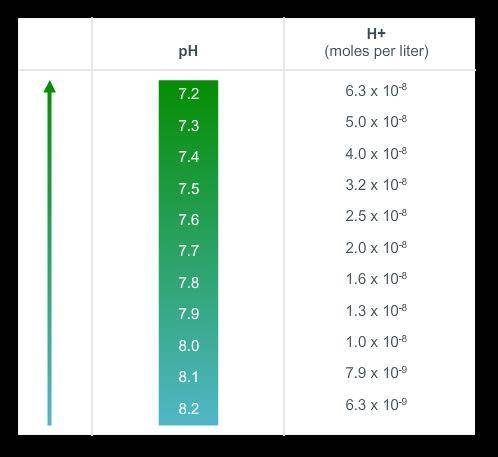
SOMEONE, please save me! The acidity of a substance relates to the proportion of free hydrogen ions (H+) it contains. Acidity is measured on a logarithmic scale. Even a small decrease in pH of ocean water can cause a large increase in hydrogen ions and acidity. This increase can have grave effects on marine ecosystems. For the last 300 million years, the average pH of the oceans has been 8.2. Today, the average pH is 8.1. Using the hydrogen ion concentrations, what is the percent increase in acidity of the ocean in the past compared to today?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:30
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 23.06.2019 05:50
Aseismic wave is energy released as the result of rock movement along a fault. t or f ?
Answers: 1

Chemistry, 23.06.2019 06:50
What is the volume of 3.2 moles of chlorine gas (cl2) at 295 k and 1.1 atm?
Answers: 1
You know the right answer?
SOMEONE, please save me!
The acidity of a substance relates to the proportion of free hydrogen ions...
Questions


History, 16.07.2019 04:00



Chemistry, 16.07.2019 04:00


Chemistry, 16.07.2019 04:00

Chemistry, 16.07.2019 04:00






Business, 16.07.2019 04:00




Social Studies, 16.07.2019 04:00

Mathematics, 16.07.2019 04:00



