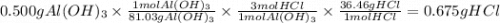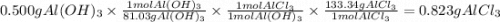Chemistry, 22.10.2020 20:01 willoughbysierra
Calculate the number of grams of HCl that can react with 0.500 g of Al(OH)3 g HCl C) Calculate the number of grams of AlCl3 formed when 0.500 g of Al(OH)3 reacts. g AlCl3

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3



Chemistry, 23.06.2019 00:40
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1
You know the right answer?
Calculate the number of grams of HCl that can react with 0.500 g of Al(OH)3 g HCl C) Calculate the n...
Questions


English, 05.11.2020 14:00

History, 05.11.2020 14:00

Mathematics, 05.11.2020 14:00



Mathematics, 05.11.2020 14:00


Mathematics, 05.11.2020 14:00

Physics, 05.11.2020 14:00

Social Studies, 05.11.2020 14:00

English, 05.11.2020 14:00



Mathematics, 05.11.2020 14:00

Social Studies, 05.11.2020 14:00

Mathematics, 05.11.2020 14:00

History, 05.11.2020 14:00