
Chemistry, 25.10.2019 18:43 panpanriser76
H2co3 is used in soft drinks. what is the name of this compound? sulfuric acid carbonic acid tartaric acid hydrochloric acid

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

Chemistry, 22.06.2019 13:30
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3
You know the right answer?
H2co3 is used in soft drinks. what is the name of this compound? sulfuric acid carbonic acid tartar...
Questions


Mathematics, 17.01.2020 12:31

English, 17.01.2020 12:31

History, 17.01.2020 12:31

Mathematics, 17.01.2020 12:31



Mathematics, 17.01.2020 12:31

Social Studies, 17.01.2020 12:31



Mathematics, 17.01.2020 12:31







History, 17.01.2020 12:31

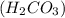 and is the reason for the formation of fizz.
and is the reason for the formation of fizz.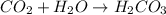
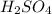 .
.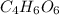
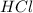 .
.

