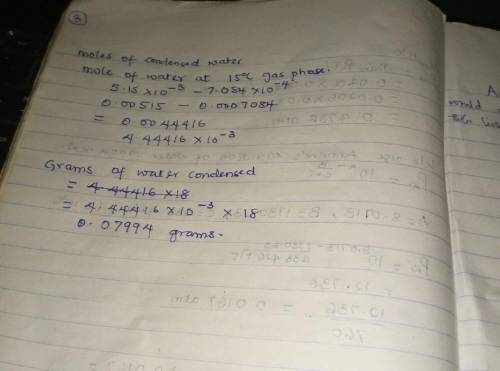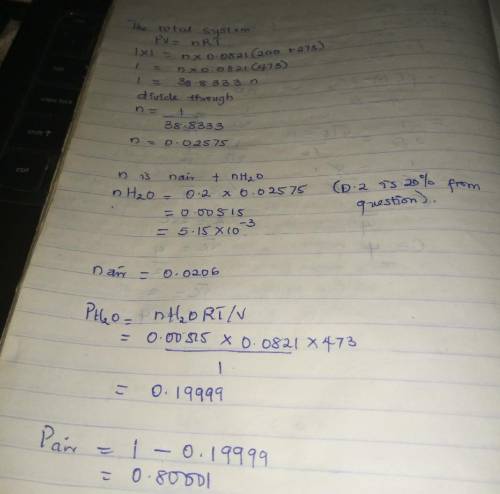Chemistry, 21.10.2020 17:01 smartgirl9989
Air containing 20.0 mol% water vapor at an initial pressure of 1 atm absolute is cooled in a 1- liter sealed vessel from 200°C to 15°C. (a) What is the pressure in the vessel at the end of the process? (Hint: The partial pressure of air in the system can be determined from the expression pair = nairRT/V and P = pair + pH2O. You may neglect the volume of the liquid water condensed, but you must show that condensation occurs.) (b) What is the mole fraction of water in the gas phase at the end of the process? (c) How much water (grams) condenses?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:20
Match the acid base pairs by arranging the acid name with the conjugate base formula. hydrogen carbonate hydrogen phosphate carbonic acid read water sulfuric acid phosphoric acid a. co32- b. hso4- c. hco3- d. po43- e. h2po4- f. oh-
Answers: 1


Chemistry, 22.06.2019 18:30
You open a can of soda at room temperature and hear a hiss. which of the following factors has changed inside the container? a.) atmospheric pressure b.) temperature of gas c.) type of gas d.) amount of gas
Answers: 1

Chemistry, 23.06.2019 00:00
What is the approximate mass of 25 cm3 of silver, if the density is 10.5 g/cm3? a. 0.42 g b. 2.4 g c. 42 g d. 260 g
Answers: 1
You know the right answer?
Air containing 20.0 mol% water vapor at an initial pressure of 1 atm absolute is cooled in a 1- lite...
Questions


Social Studies, 19.05.2021 06:10


French, 19.05.2021 06:10




Mathematics, 19.05.2021 06:10

Mathematics, 19.05.2021 06:10




English, 19.05.2021 06:10

Mathematics, 19.05.2021 06:10

Mathematics, 19.05.2021 06:10


Mathematics, 19.05.2021 06:10

History, 19.05.2021 06:10

Mathematics, 19.05.2021 06:10