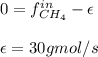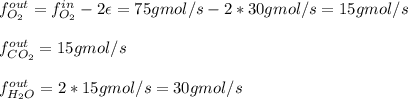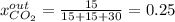Chemistry, 21.10.2020 16:01 HaileyAnn04
For the combustion of methane presented in Example 5.4, the chemical reaction is CH4 +2O2 →CO2 +2H2O Suppose that methane flows into a burner at 30 gmol/s, while oxygen flows into the same burner at 75 gmol/s. If all the meth- ane is burned and a single output stream leaves the burner, what is the mole fraction of CO2 in that output stream? Hint 1: Does the fact that all the methane is burned mean that all the oxygen is burned also? Hint 2: Find the molar flow rate of each component gas in the outlet gas ("flue gas").

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 10:30
How do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 2

Chemistry, 22.06.2019 15:40
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

Chemistry, 22.06.2019 19:00
How many moles of cu are needed to react with 5.8 moles of agno3? cu + 2 agno3 → cu(no3)2 + 2 ag
Answers: 3
You know the right answer?
For the combustion of methane presented in Example 5.4, the chemical reaction is CH4 +2O2 →CO2 +2H2O...
Questions



History, 27.07.2021 23:40

Mathematics, 27.07.2021 23:40









Computers and Technology, 27.07.2021 23:40

Computers and Technology, 27.07.2021 23:40

Mathematics, 27.07.2021 23:40

Mathematics, 27.07.2021 23:40


Mathematics, 27.07.2021 23:40


Mathematics, 27.07.2021 23:40

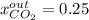
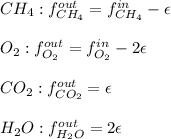
 accounts for the reaction extent. However, as all the methane is consumed, from the methane balance:
accounts for the reaction extent. However, as all the methane is consumed, from the methane balance: