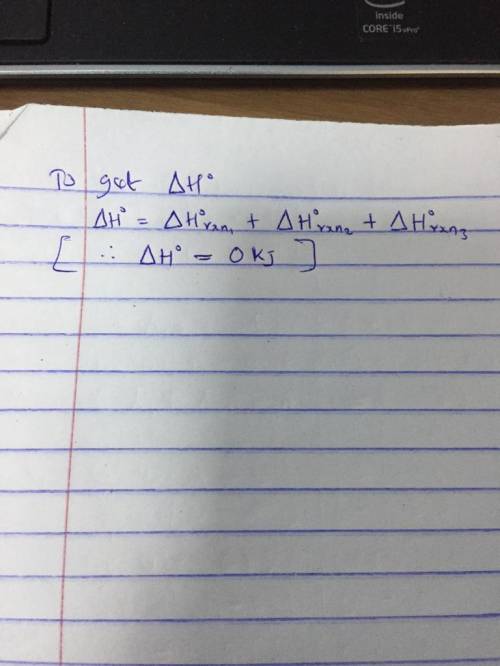6.82
A 2.10-mole sample of crystalline acetic acid, ini-
tially at 17.0°C, is allowed t...

6.82
A 2.10-mole sample of crystalline acetic acid, ini-
tially at 17.0°C, is allowed to melt at 17.0°C and is
then heated to 118.1°C (its normal boiling point) at
1.00 atm. The sample is allowed to vaporize at.
118.1°C and is then rapidly quenched to 17.0°C, so
that it recrystallizes. Calculate AH° for the total pro-
cess as described.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:50
Which best describes why nh4+ can form an ionic bond with cl-?
Answers: 3

Chemistry, 21.06.2019 18:00
Aballoon inflated with three breaths of air has a volume of 1.7 l. at the same temperature and pressure, what is the volume of the balloon if five more same-sized breaths are added to the balloon?
Answers: 3

Chemistry, 21.06.2019 21:30
Balance this equation co2(g) + h2o (g) show that the balanced equation obeys the law if conversation of mass
Answers: 1

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3
You know the right answer?
Questions


Social Studies, 20.08.2020 05:01




Mathematics, 20.08.2020 05:01

Geography, 20.08.2020 05:01


Mathematics, 20.08.2020 05:01


Social Studies, 20.08.2020 05:01


Engineering, 20.08.2020 05:01

Social Studies, 20.08.2020 05:01


Spanish, 20.08.2020 05:01



Chemistry, 20.08.2020 05:01