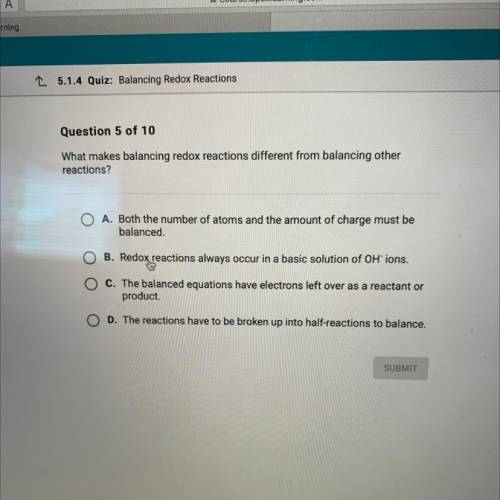
What makes balancing redox reactions different from balancing other
reactions?
A. Both the nu...

What makes balancing redox reactions different from balancing other
reactions?
A. Both the number of atoms and the amount of charge must be
balanced.
B. Redox reactions always occur in a basic solution of OH ions.
ChThe balanced equations have electrons left over as a reactant or
"product.
D. The reactions have to be broken up into half-reactions to balance.
SUBMIT

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1

Chemistry, 22.06.2019 03:30
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 22.06.2019 18:00
Which statement best describes the he properties of iconic compounds ?
Answers: 1
You know the right answer?
Questions

Computers and Technology, 30.07.2021 17:30

Mathematics, 30.07.2021 17:30


Biology, 30.07.2021 17:30




Mathematics, 30.07.2021 17:30

Computers and Technology, 30.07.2021 17:30






Computers and Technology, 30.07.2021 17:30


Mathematics, 30.07.2021 17:30

Computers and Technology, 30.07.2021 17:30





