

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 21:20
One way in which the useful metal copper is produced is by dissolving the mineral azurite, which contains copper(ii) carbonate, in concentrated sulfuric acid. the sulfuric acid reacts with the copper(ii) carbonate to produce a blue solution of copper(ii) sulfate. scrap iron is then added to this solution, and pure copper metal precipitates out because of the following chemical reaction: (s) (aq) (s) (aq) suppose an industrial quality-control chemist analyzes a sample from a copper processing plant in the following way. he adds powdered iron to a copper(ii) sulfate sample from the plant until no more copper will precipitate. he then washes, dries, and weighs the precipitate, and finds that it has a mass of .
Answers: 2

Chemistry, 22.06.2019 23:10
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium.b)heavier than helium.c)the same weight as helium.d)dependent on the element that reacted with carbon.
Answers: 3


Chemistry, 23.06.2019 04:20
The graph shows one consequence of urban sprawl. how did urban sprawl contribute to the change in biodiversity
Answers: 2
You know the right answer?
0.90 g of hydrogen chloride (HCl) is dissolved in water to make 2.0 L of solution. What is the pH of...
Questions

Social Studies, 25.06.2019 09:30

History, 25.06.2019 09:30

Computers and Technology, 25.06.2019 09:30


Computers and Technology, 25.06.2019 09:30







Mathematics, 25.06.2019 09:30

Advanced Placement (AP), 25.06.2019 09:30

Mathematics, 25.06.2019 09:30

Mathematics, 25.06.2019 09:30



History, 25.06.2019 09:30


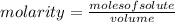
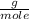 , then the following rule of three can be applied: if there are 35.45 grams of HCl in 1 mole, 0.9 grams of HCl in how many moles will they be?
, then the following rule of three can be applied: if there are 35.45 grams of HCl in 1 mole, 0.9 grams of HCl in how many moles will they be?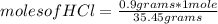
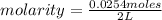
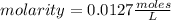
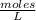 . So [HCl] = [H⁺]
= 0.0127
. So [HCl] = [H⁺]
= 0.0127 


