
Chemistry, 20.10.2020 16:01 Flameking1223
Aspirin is a weak organic acid whose molecular formula is HC9H7O4. An aqueous solution of aspirin is prepared by dissolving 3.60 g/L. The pH of this solution is found to be 2.6. Calculate Ka for aspirin. (atomic mass: C

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:00
What is the empirical formula of vanadium 1 oxide given that 20.38 grams of vandium combines with oxygen to form 23.58 grams of the oxide
Answers: 1

Chemistry, 22.06.2019 18:30
When the chemicals iron sulfide (fes) and hydrochloric acid (hcl) are combined, bubbles appear from the mixture. 1. does the appearance of bubbles indicate a physical or chemical change? 2. why do the bubbles indicate this change? 3. what property is this?
Answers: 1

Chemistry, 22.06.2019 21:00
Read "who built the pyramids? ”. leave this link open while you answer the questions throughout the assignment. give at least two reasons why some people claim the pyramids of giza were constructed by aliens.
Answers: 1

Chemistry, 23.06.2019 10:00
Abike ride event is 30 miles. a first aid tent is put at the 3/4 mark of the course. how many miles from the starting point is the first aid tent?
Answers: 1
You know the right answer?
Aspirin is a weak organic acid whose molecular formula is HC9H7O4. An aqueous solution of aspirin is...
Questions

Mathematics, 21.08.2021 18:10

Mathematics, 21.08.2021 18:20


English, 21.08.2021 18:20




Physics, 21.08.2021 18:20


Chemistry, 21.08.2021 18:20

History, 21.08.2021 18:20

English, 21.08.2021 18:20



Mathematics, 21.08.2021 18:20

Arts, 21.08.2021 18:20

Social Studies, 21.08.2021 18:20


English, 21.08.2021 18:20

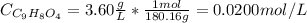
![Ka = \frac{[C_{9}H_{7}O_{4}^{-}][H_{3}O^{+}]}{[C_{9}H_{8}O_{4}]}](/tpl/images/0824/0933/a1a8e.png)
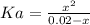
![pH = -log[H_{3}O^{+}]](/tpl/images/0824/0933/b7638.png)
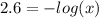
![x = 2.51 \cdot 10^{-3} M = [H_{3}O^{+}] = [C_{9}H_{7}O_{4}^{-}]](/tpl/images/0824/0933/98904.png)
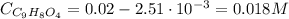
![Ka = \frac{[C_{9}H_{7}O_{4}^{-}][H_{3}O^{+}]}{[C_{9}H_{8}O_{4}]} = \frac{(2.51 \cdot 10^{-3})^{2}}{0.018} = 3.50 \cdot 10^{-4}](/tpl/images/0824/0933/393ec.png)


