Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 13:30
Some animals that try to adapt to climate changes eventually die due to starvation, as climate change alters the web.
Answers: 2

Chemistry, 22.06.2019 18:30
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1

Chemistry, 22.06.2019 19:00
How does kepler second law of planetary motion overthrow one of the basic beliefs of classical astronomy
Answers: 1
You know the right answer?
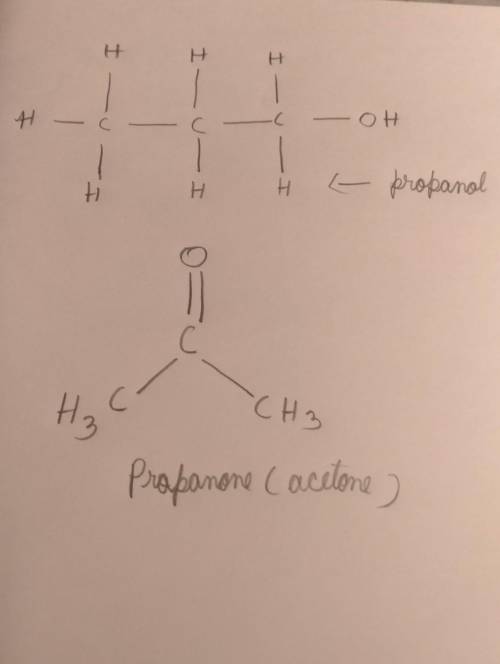
2 propanoate on oxidation gives ?...
Questions



Mathematics, 20.10.2020 04:01

Mathematics, 20.10.2020 04:01

Mathematics, 20.10.2020 04:01





Mathematics, 20.10.2020 04:01

History, 20.10.2020 04:01

Physics, 20.10.2020 04:01



Chemistry, 20.10.2020 04:01



Mathematics, 20.10.2020 04:01