
Chemistry, 17.10.2020 14:01 loran55667
Is someone could please help me answer 6-12 on this I will greatly appreciate it, please and thank you
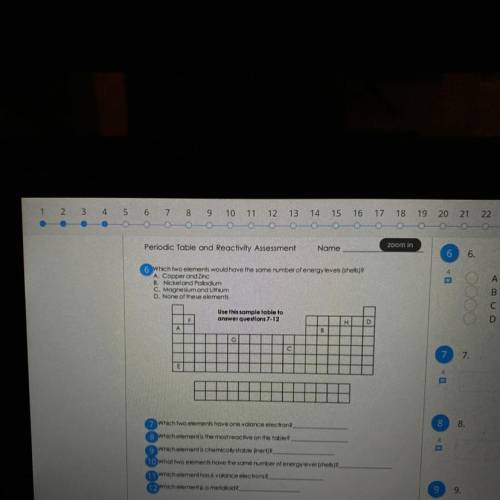

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:20
An aqueous solution of calcium hydroxide is standardized by titration with a 0.120 m solution of hydrobromic acid. if 16.5 ml of base are required to neutralize 27.5 ml of the acid, what is the molarity of the calcium hydroxide solution?
Answers: 3

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1

Chemistry, 22.06.2019 11:00
When hydrochloric acid reacts with potassium hydroxide solution, the following reaction occurs. hcl (aq) + koh (aq) h2o (l) + kcl (aq) the reaction gives off heat energy, so it is an reaction.
Answers: 1
You know the right answer?
Is someone could please help me answer 6-12 on this I will greatly appreciate it, please and thank y...
Questions



History, 13.06.2021 20:30



Engineering, 13.06.2021 20:30


Chemistry, 13.06.2021 20:30

Computers and Technology, 13.06.2021 20:30

Social Studies, 13.06.2021 20:30


History, 13.06.2021 20:30

English, 13.06.2021 20:40

Biology, 13.06.2021 20:40

Health, 13.06.2021 20:40

English, 13.06.2021 20:40

Social Studies, 13.06.2021 20:40

Mathematics, 13.06.2021 20:40


Mathematics, 13.06.2021 20:40



