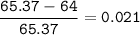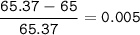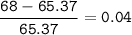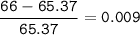Zinc has an average atomic mass of 65.37 amu. Jack is trying to figure out what the most abundant isotope of zinc is, but he doesn't have access to the internet, so the average atomic mass is the only information he has. Jack decides to made an educated guess as to the most abundant isotope of zinc by assuming there are only two isotopes of zinc. If Jack assumes there are only two isotopes of Zn, what he be most likely to decide is zinc's most abundant isotope?
Question 5 options:
Zinc-64
Zinc-65
Zinc-68
Zinc-66

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:40
Describe in detail the melting point behavior of the 80: 20 benzoic acid-mandelic acid mixture
Answers: 3

Chemistry, 22.06.2019 00:00
How many liters of water vapor can be produced if 108 grams of methane gas (ch4) are combusted at 312 k and 0.98 atm? show all work. pls ! will mark as brainliest
Answers: 1

Chemistry, 22.06.2019 21:30
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1

You know the right answer?
Zinc has an average atomic mass of 65.37 amu. Jack is trying to figure out what the most abundant is...
Questions

English, 26.09.2019 09:30

Chemistry, 26.09.2019 09:30

Mathematics, 26.09.2019 09:30


Mathematics, 26.09.2019 09:30

History, 26.09.2019 09:30






Health, 26.09.2019 09:30


Social Studies, 26.09.2019 09:30



History, 26.09.2019 09:30

History, 26.09.2019 09:30