
Chemistry, 16.10.2020 05:01 am2garcia5
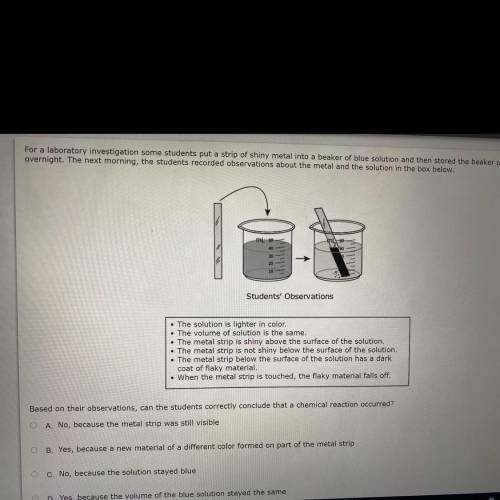
For a laboratory investigation some students put a strip of shiny metal into a beaker of blue solution and then stored the beaker on a shelf
overnight. The next morning, the students recorded observations about the metal and the solution in the box below.
Students' Observations
Exhibit
. The solution is lighter in color,
The volume of solution is the same.
• The metal strip is shiny above the surface of the solution
• The metal strip is not shiny below the surface of the solution.
• The metal strip below the surface of the solution has a dark
coat of flaky material
• When the metal strip is touched, the flaky material falls off.
Based on their observations, can the students correctly conclude that a chemical reaction occurred?
A. No, because the metal strip was still visible
B. Yes, because a new material of a different color formed on part of the metal strip
c. No, because the solution stayed blue
ble solution stayed the same
12 AM

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which supports the idea that birds and butterflies both have wings but they do not have a common ancestor with wings? a. the wings are analogous structures that evolved differently and do not have a similar internal structure. b. the wings are homologous structures that evolved differently and do not have a similar internal structure. c. wings of birds are vestigial structures, but the wing structures of bats are not vestigial. d. wings of bats are vestigial structures, but the wing structures of birds are not vestigial
Answers: 1

Chemistry, 22.06.2019 01:30
Sulfuric acid (a component of acid rain) reacts with limestone (calcium carbonate) to produce calcium sulfate and carbon dioxide. this damages buildings and statues made of limestone. which solution of sulfuric acid will damage these structures more quickly? a. 0.001% b. 0.005% c. 0.010% d. 0.015%
Answers: 3

Chemistry, 22.06.2019 03:40
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and gas called? identify which way they are transitioning
Answers: 2
You know the right answer?
For a laboratory investigation some students put a strip of shiny metal into a beaker of blue soluti...
Questions


Chemistry, 19.09.2019 02:30


Chemistry, 19.09.2019 02:30




Mathematics, 19.09.2019 02:30




Mathematics, 19.09.2019 02:30



Mathematics, 19.09.2019 02:30

Mathematics, 19.09.2019 02:30

Chemistry, 19.09.2019 02:30


Biology, 19.09.2019 02:30

Mathematics, 19.09.2019 02:30



