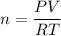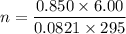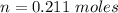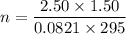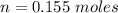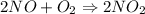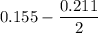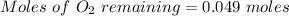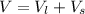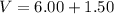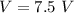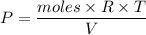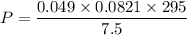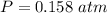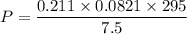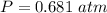Two bulbs are connected by a stopcock. The large bulb, with a volume of 6.00 L, contains nitric oxide at a pressure of 0.850 atm, and the small bulb, with a volume of 1.50 L, contains oxygen at a pressure of 2.50 atm. The temperature at the beginning and the end of the experiment is 22∘C .
After the stopcock is opened, the gases mix and react.
2NO(g)+O2(g)→2NO2(g)
1. Which gases are present at the end of the experiment?
2. What are the partial pressures of the gases? If the gas was consumed completely, put 0 for the answer.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
The drawing represents the movement of particles in a substance. what changes of state can this substance undergo
Answers: 1

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1

Chemistry, 22.06.2019 12:30
Consider the four elements above. which one of these elements will combine with oxygen in a 1: 1 ratio?
Answers: 3

Chemistry, 22.06.2019 14:00
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1
You know the right answer?
Two bulbs are connected by a stopcock. The large bulb, with a volume of 6.00 L, contains nitric oxid...
Questions




English, 06.01.2020 16:31


Biology, 06.01.2020 16:31

Arts, 06.01.2020 16:31

Mathematics, 06.01.2020 16:31

Mathematics, 06.01.2020 16:31

Mathematics, 06.01.2020 16:31


Mathematics, 06.01.2020 16:31

Mathematics, 06.01.2020 16:31

Mathematics, 06.01.2020 16:31

Mathematics, 06.01.2020 16:31


Physics, 06.01.2020 16:31


Mathematics, 06.01.2020 16:31

Mathematics, 06.01.2020 16:31