
Chemistry, 14.10.2020 14:01 adriannacomrosenbark
The rate of reaction in terms of the "rate law expression" includes the rate constant (kk), the concentration of the reactants, and the orders of the reaction with respect to the different reactants. Consider the following reaction: A+B→C+DA+B→C+D The initial concentrations of the reactants AA and BB are 0.200 MM and 0.350 MM, respectively. The rate of reaction is 0.060 M⋅s−1M⋅s−1, and the orders of the reaction, with respect to reactants AA and BB, are 1 and 2, respectively. Determine the rate constant (kk) for the reaction using the rate law. Express your answer in M−2⋅s−1M−2⋅s−1 to three significant figures.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:40
During which time interval does the object travel approximately 10 meters
Answers: 3



Chemistry, 22.06.2019 15:30
Draw the lewis dot structure for each of the following polyatomic ions
Answers: 1
You know the right answer?
The rate of reaction in terms of the "rate law expression" includes the rate constant (kk), the conc...
Questions

Mathematics, 30.11.2020 22:10




Biology, 30.11.2020 22:10

Mathematics, 30.11.2020 22:10






English, 30.11.2020 22:10

Chemistry, 30.11.2020 22:10







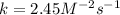
![r=k[A][B]^2](/tpl/images/0805/6865/487f0.png)
![k=\frac{r}{[A][B]^2}\\ \\k=\frac{0.060M/s}{(0.200M)(0.350M)^2} \\\\k=2.45M^{-2}s^{-1}](/tpl/images/0805/6865/72630.png)


