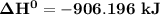Chemistry, 13.10.2020 03:01 giraffesaur44
The first reaction in the Ostwald process for the production of nitric acid involves the combustion of ammonia
4NH3(g)+5O2(g)=4NO(g)+6H2O(g)
a) Estimate ΔH^o (in kJ) for this reaction using average bond energies.
b) Calculate ΔH^o (in kJ) for this reaction using standard heats of formation.
c) Briefly explain why the value for ΔH^o, calculated using average bond energies, is only considered to be an estimate of the standard enthalpy change for the reaction

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Apiece of metal with a length of 1.42 cm was measured using four different instruments. which of the following measurements is the most accurate?
Answers: 3


Chemistry, 23.06.2019 02:40
How can a mixture of salt water be separated into salt and water
Answers: 1

Chemistry, 23.06.2019 04:40
[01.07]what is the answer to the problem: 101 g + 25.01 g + 5.05 g? 131.06 g 131.1 g 131 g 130 g
Answers: 1
You know the right answer?
The first reaction in the Ostwald process for the production of nitric acid involves the combustion...
Questions

English, 27.08.2019 19:30



Biology, 27.08.2019 19:30

Social Studies, 27.08.2019 19:30

Chemistry, 27.08.2019 19:30



Mathematics, 27.08.2019 19:30


Social Studies, 27.08.2019 19:30

Mathematics, 27.08.2019 19:30


Computers and Technology, 27.08.2019 19:30






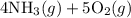 ↔
↔ 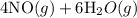
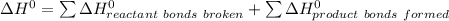
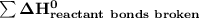 = 7182 kJ
= 7182 kJ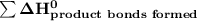 = - 8032 kJ
= - 8032 kJ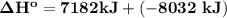

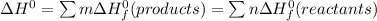
![\Delta H^0 = [ 4 \ mol \times \Delta H^0_f \ (NO(g)) + 6\ mol \times \Delta H^0_f(H_2O)] - [ 4 \ mol \times \Delta H^0_f \ (NH_3(g)) + 5 \ mol \times \Delta H^0_f \ (O_2)]](/tpl/images/0802/3228/2e247.png)
![\Delta H^0 = [ 4 \ mol \times90.29 \ kJ/mol + 6\ mol \times -241.826 \ kJ/mol - [ 4 \ mol \times-45.9 \ kJ/mol + 5 \ mol \times 0]](/tpl/images/0802/3228/35715.png)