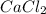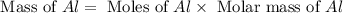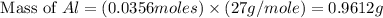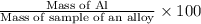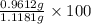5. A 1.1181-g sample of an alloy (a mixture) of aluminum and magnesium was treated with an
excess of sodium hydroxide solution. In the reaction, only the aluminum reacts with the sodium
hydroxide solution:
2 Al + 2 NaOH + 6 H202 Na[Al(OH)4] + 3 H2
If 0.1068 g of H2 is produced, what is the mass percent of aluminum in the alloy?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:30
A48 g piece of ice at 0.0 ∘c is added to a sample of water at 7.4 ∘c. all of the ice melts and the temperature of the water decreases to 0.0 ∘c. how many grams of water were in the sample?
Answers: 1

Chemistry, 21.06.2019 14:30
An occluded front moves over the farmland that has been experiencing drought conditions. what change in weather will this front likely bring? a. gray skies, but no rain b. an extended period of rain c. more dry air and sunny skies d. violent, short-lived thunderstorms
Answers: 3

Chemistry, 22.06.2019 06:30
What is the correct term for living the most sustainable life you can within your current circumstances?
Answers: 1

Chemistry, 23.06.2019 00:00
Which is true about metals used for jewelry, such as platinum and gold? a. they have low flammability. b. they have low reactivity. c. they have high flammability. d. they have high reactivity.
Answers: 1
You know the right answer?
5. A 1.1181-g sample of an alloy (a mixture) of aluminum and magnesium was treated with an
excess o...
Questions




Mathematics, 02.02.2021 17:00





Mathematics, 02.02.2021 17:00

Mathematics, 02.02.2021 17:00

Advanced Placement (AP), 02.02.2021 17:00

English, 02.02.2021 17:00

Mathematics, 02.02.2021 17:10


Mathematics, 02.02.2021 17:10


Mathematics, 02.02.2021 17:10


Mathematics, 02.02.2021 17:10

Mathematics, 02.02.2021 17:10

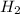 = 0.1068 g
= 0.1068 g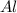 = 27 g/mol
= 27 g/mol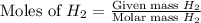
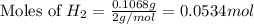
![2Al+2NaOH+6H_2O\rightarrow 2Na[Al(OH)_4]+3H_2](/tpl/images/0796/4861/bae6d.png)
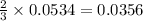 mole of
mole of