
Chemistry, 02.10.2020 19:01 angoliabirtio
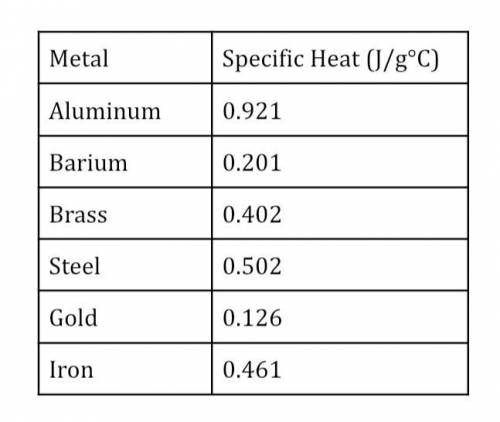
An unknown metal with the mass of 10.0g is heated from 25.0°C to 87.2°C with 250.0 joules of heat. Calculate specific heat (Cp) and then use the chart of metals and their specific heat to determine the identity of this metal.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1

Chemistry, 22.06.2019 14:30
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1

Chemistry, 22.06.2019 17:30
In a heat of an engine, if 700 j enters the system, and the piston does 400 j of work what is the final internal (thermal) energy of the system if the initial energy is 1,500 j
Answers: 2
You know the right answer?
An unknown metal with the mass of 10.0g is heated from 25.0°C to 87.2°C with 250.0 joules of heat. C...
Questions

English, 19.02.2021 02:00

Mathematics, 19.02.2021 02:00





History, 19.02.2021 02:00


Mathematics, 19.02.2021 02:00


Mathematics, 19.02.2021 02:00


Mathematics, 19.02.2021 02:00



Mathematics, 19.02.2021 02:00

Mathematics, 19.02.2021 02:00

Chemistry, 19.02.2021 02:00

English, 19.02.2021 02:00

Mathematics, 19.02.2021 02:00



