
Chemistry, 24.09.2020 15:01 yungking1329
Calculate the molar volume occupied by 1 mole of N2 using the van der Waals equation in the form of virial expansion at (a) its critical temperature and (b) its Boyle temperature. Assume that the pressure is 10 atm throughout. At what temperature is the gas most perfect? Use the following data: Tc = 126.3 K, a=1.352 L2 atm mol-2, b = 0.0387 L mol-1

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Different isotopes indicate that an element will have different numbers of
Answers: 2

Chemistry, 22.06.2019 09:00
Which process does not require the presence of a physical substance in order to transfer heat? air in the atmosphere is heated by the ground. this warm air then rises, and cooler air falls. this is an example of what type of process? how is conduction different from radiation?
Answers: 1

Chemistry, 22.06.2019 11:40
Consider this equilibrium: n29) + o2(g) + 2no(c).nitrogen gas and oxygen gas react when placed in a closed container. as the reaction proceeds towards equilibrium, what happens to the rate of thereverse reaction?
Answers: 1

Chemistry, 22.06.2019 14:30
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2
You know the right answer?
Calculate the molar volume occupied by 1 mole of N2 using the van der Waals equation in the form of...
Questions


English, 23.06.2019 16:00

Mathematics, 23.06.2019 16:00

Mathematics, 23.06.2019 16:00




History, 23.06.2019 16:00

Health, 23.06.2019 16:00


Biology, 23.06.2019 16:00





Mathematics, 23.06.2019 16:00



History, 23.06.2019 16:00

History, 23.06.2019 16:00

 = 1 mole
= 1 mole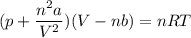
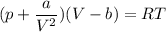
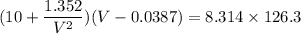
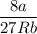
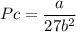
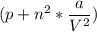 depending on the circumstances.
depending on the circumstances.

