
Chemistry, 20.09.2020 15:01 smithsavannah295
olecular iodine, I2(g), dissociates into iodine atoms at 625 K with a first-order rate constant of 0.271 s-1. (a) What is the half-life for this reaction? s (b) If you start with 0.048 M I2 at this temperature, how much will remain after 5.37 s assuming that the iodine atoms do not recombine to form I2? M

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 18:30
The famous scientist galileo galilei did several experiments with sloping planes, which he rolled metal balls down so that he could study motion. by changing the slope, he could study how the speed at which the ball rolled was affected. what was the independent variable in galileo's experiment? a. the speed of the ball b. the slope of the plane c. whether the ball moved d. what the ball was made of
Answers: 2

Chemistry, 22.06.2019 22:30
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1
You know the right answer?
olecular iodine, I2(g), dissociates into iodine atoms at 625 K with a first-order rate constant of 0...
Questions

History, 05.05.2020 18:02



Business, 05.05.2020 18:02

Engineering, 05.05.2020 18:02




English, 05.05.2020 18:02

History, 05.05.2020 18:02





Biology, 05.05.2020 18:02

English, 05.05.2020 18:02


English, 05.05.2020 18:02


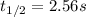
![[I_2]=0.011M](/tpl/images/0771/9802/19391.png)
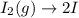
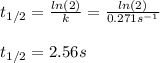
![[I_2]=[I_2]_0exp(-kt)](/tpl/images/0771/9802/e9e96.png)
![[I_2]=0.048Mexp(-0.271s^{-1}*5.37s)\\\\](/tpl/images/0771/9802/4a6e2.png)


