
Chemistry, 20.09.2020 17:01 kathrynaveda
The first-order rate constant for the decomposition of N2O5, given below, at 70°C is 6.82 10-3 s-1. Suppose we start with 0.0200 mol of N2O5(g) in a volume of 2.5 L. 2 N2O5(g) → 4 NO2(g) + O2(g) (a) How many moles of N2O5 will remain after 2.5 min? mol (b) How many minutes will it take for the quantity of N2O5 to drop to 0.005 mol? min (c) What is the half-life of N2O5 at 70°C?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:10
56.16 gregor mendel was the first scientist to use statistics to analyze scientific data. before mendel's experiments, scientists believed that organisms acquired traits from their environment and passed them on to their offspring. after mendel's discoveries were accepted, scientists realized that traits passed to offspring were the result of genes being passed from parents to offspring. this is an example of pls
Answers: 1

Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

Chemistry, 22.06.2019 18:10
Areader can tell that the meaning of “obnoxious” will include “having the quality of something” because of the .a) prefix b)pronunciation c)suffix d) word root
Answers: 3

Chemistry, 22.06.2019 18:30
Which of the following nuclei would be the least stable a 2 protons, 2 neutrons b 1 proton 1 neutron c 1 proton 3 neutrons d 1 proton 2 neutrons
Answers: 3
You know the right answer?
The first-order rate constant for the decomposition of N2O5, given below, at 70°C is 6.82 10-3 s-1....
Questions

Computers and Technology, 24.02.2020 18:55




Mathematics, 24.02.2020 18:55




English, 24.02.2020 18:55










Physics, 24.02.2020 18:55

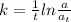
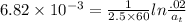
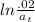 = 1.023
= 1.023 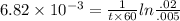
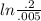 = 3.689
= 3.689


