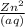or the solution containing Zn2+(aq), n is 2, so the MM/n ratio will be the molar mass of Zn divided by 2: MM/n = (65.39 g/mol) / 2 = 32.70 g/mol Compare this theoretical value with your experimental result. If the plating efficiency is significantly less than 1, say it is 0.55, how would the observed MM/n ratio compare to the theoretical value?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 16:30
How many atoms of oxygen are contained in 160 grams of n2o3
Answers: 2

Chemistry, 21.06.2019 23:00
Select the correct answer. given: 2libr + ba → babr2 + 2li in this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced? a. 1.18 mol b. 2.37 mol c. 4.73 mol d. 16.4 mol e. 32.9 mol
Answers: 2

Chemistry, 22.06.2019 01:30
When an object falls through the air and encounters air resistance its overall speed will be than if it had not encountered air resistance? (one word answer)
Answers: 2

Chemistry, 22.06.2019 02:30
You have a sample of a gas that occupies a volume of 17ml at -111 degrees celsius. what volume does the sample occupy at 88 degrees celsius? show all work asap
Answers: 3
You know the right answer?
or the solution containing Zn2+(aq), n is 2, so the MM/n ratio will be the molar mass of Zn divided...
Questions





Mathematics, 26.03.2021 02:00

Mathematics, 26.03.2021 02:00


English, 26.03.2021 02:00

Mathematics, 26.03.2021 02:00

Mathematics, 26.03.2021 02:00


History, 26.03.2021 02:00

Biology, 26.03.2021 02:00

History, 26.03.2021 02:00



Mathematics, 26.03.2021 02:00