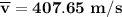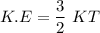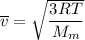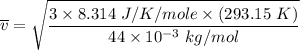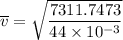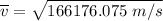Consider a closed container of gas that is a mixture of 30% CO2 and 70% N2 at room temperature 20°C. The gases are in thermal equilibrium with one another. a) Which has the higher kinetic energy, the average CO2 or N2 molecule? b) What is that root-mean-square velocity of a CO2 molecule? For reference, a carbon atom has 6 protons, a nitrogen atom has 7 protons, and an oxygen atom has 8 protons.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Different isotopes indicate that an element will have different numbers of
Answers: 2

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and gas called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 23:00
Arectangle has a diagonal 20 inches long that forms angles of 60 and 30 with the sides. find the perimeter of the rectangle. for geometry
Answers: 3

Chemistry, 23.06.2019 01:00
What is the chemical name of the compound ti2o3? use the list of polyatomic ions and the periodic table to you answer.
Answers: 1
You know the right answer?
Consider a closed container of gas that is a mixture of 30% CO2 and 70% N2 at room temperature 20°C....
Questions


Mathematics, 03.03.2020 21:55

Mathematics, 03.03.2020 21:55

Mathematics, 03.03.2020 21:55




Biology, 03.03.2020 21:55

History, 03.03.2020 21:56

English, 03.03.2020 21:56

English, 03.03.2020 21:56

Biology, 03.03.2020 21:56

Mathematics, 03.03.2020 21:56


History, 03.03.2020 21:56





English, 03.03.2020 21:56