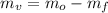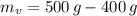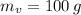Chemistry, 07.09.2020 05:01 mqturner1989Kedie
En un vaso de precipitado de un litro se coloca exactamente 500 mL de agua destilada a temperatura ambiente y se realizan dos experimentos.
a) Primero: Se congela a 0°C y se observa que su volumen aumenta.
b) Segundo: Se evapora durante el día y se observa que su volumen disminuye a 400 mL.
Preguntas:
1. ¿Cuál es la masa de agua a temperatura ambiente?
2. ¿Cuál es la masa de agua cuando se congela?
3. ¿Cuál es la masa de agua que queda después de la evaporación?
4. ¿Qué masa de agua se evaporó?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
When an object falls through the air and encounters air resistance its overall speed will be than if it had not encountered air resistance? (one word answer)
Answers: 2

Chemistry, 22.06.2019 05:30
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1


Chemistry, 22.06.2019 12:30
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1
You know the right answer?
En un vaso de precipitado de un litro se coloca exactamente 500 mL de agua destilada a temperatura a...
Questions








Mathematics, 01.07.2020 19:01


Mathematics, 01.07.2020 19:01



English, 01.07.2020 19:01


Computers and Technology, 01.07.2020 19:01

English, 01.07.2020 19:01

Mathematics, 01.07.2020 19:01

Mathematics, 01.07.2020 19:01

Mathematics, 01.07.2020 19:01

History, 01.07.2020 19:01

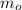 ), medido en gramos, al conocer su densidad (
), medido en gramos, al conocer su densidad (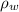 ), medida en gramos por mililitro, y volumen inicial ocupado en el vaso de precipitado (
), medida en gramos por mililitro, y volumen inicial ocupado en el vaso de precipitado (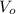 ), medido en mililitros, a partir de la siguiente expresión:
), medido en mililitros, a partir de la siguiente expresión: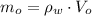
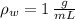 y
y 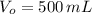 , entonces:
, entonces: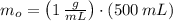
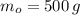
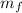 ), medido en gramos, de la sustancia al multiplicar el volumen final (
), medido en gramos, de la sustancia al multiplicar el volumen final (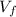 ), medido en mililitros, por la densidad del agua (
), medido en mililitros, por la densidad del agua (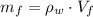
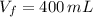 , entonces:
, entonces: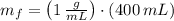
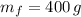
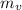 ), medida en gramos, es igual a la diferencia entre las masas inicial y final, ambas medidas en gramos:
), medida en gramos, es igual a la diferencia entre las masas inicial y final, ambas medidas en gramos: