
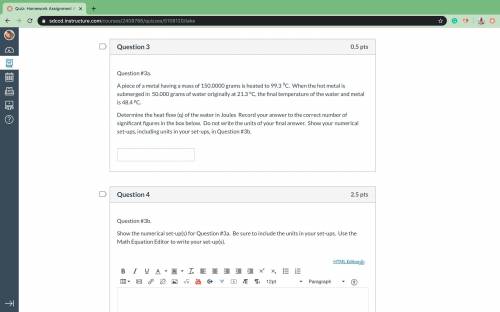
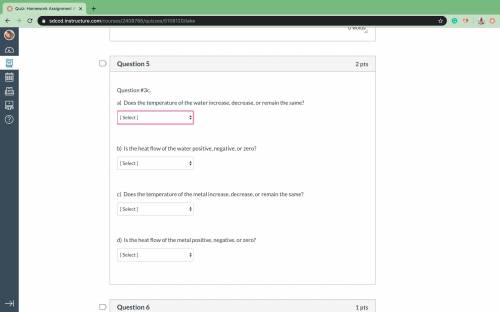
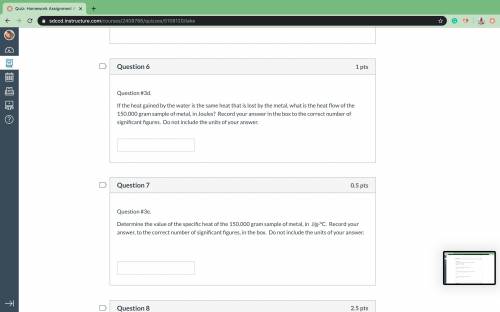
A piece of a metal having a mass of 150.0000 grams is heated to 99.3 9C. When the hot metal is submerged in 50.000 grams of water originally at 21.3 oC, the final temperature of the water and metal is 48.4 pC. Determine the heat flow (q) of the water in Joules Record your answer to the correct number of significant figures

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Plz mark brainliest 30 points1) find the momentum of a 12 kg snowball that is rolling with a velocity of 9 m/s.2) an 8 ball with a mass of .5 kg is sitting at rest. it is hit by the cue ball (1 kg) traveling at 2.5 m/s. if the cue ball is at rest after the collision, how fast is the 8 ball traveling after the collision? 3) two football players are running toward each other. if the offensive player is 75 kg and is running 8 m/s, how fast must the 60 kg defensive player run in order for the two players to hit and stop?
Answers: 1

Chemistry, 22.06.2019 17:30
Upon decomposition, one sample of magnesium fluoride produced 1.65 kg of magnesium and 2.56 kg of fluorine. a second sample produced 1.32 kg of magnesium. part a how much fluorine (in grams) did the second sample produce?
Answers: 2

Chemistry, 23.06.2019 00:30
The footprints of a dinosaur and the burrow of an ancient shrimp are examples of which kind of fossils
Answers: 2

You know the right answer?
A piece of a metal having a mass of 150.0000 grams is heated to 99.3 9C. When the hot metal is subme...
Questions






Mathematics, 19.06.2020 18:57



Mathematics, 19.06.2020 18:57




English, 19.06.2020 18:57







Mathematics, 19.06.2020 18:57



