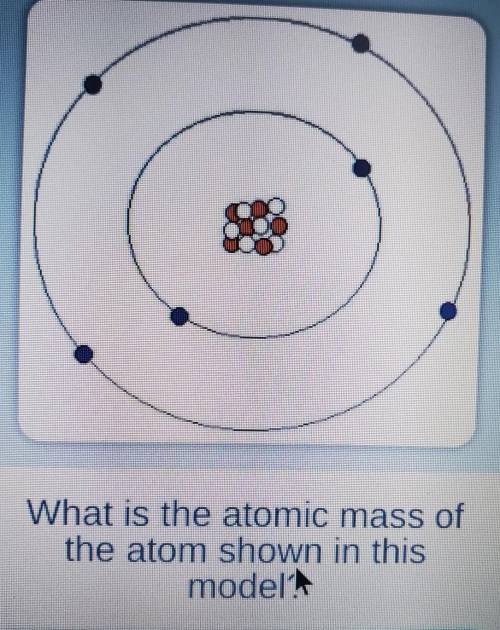
What is the atomic mass of the atom shown in this model
...


Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

Chemistry, 22.06.2019 08:40
Write the formula for the following chemicals. 7. e. trinitrogen tetraoxide a calcium phosphate f. magnesium acetate b. potassium sulfide g nickel(iii) cyanide c carbon dioxide h. silver sulfate d. cobalt(ii) chloride
Answers: 1

Chemistry, 22.06.2019 10:50
An atom of lithium-7 has an equal number of(1) electrons and neutrons(2) electrons and protons(3) positrons and neutrons(4) positrons and protons
Answers: 2

Chemistry, 22.06.2019 16:30
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u.s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2
You know the right answer?
Questions

Health, 10.11.2020 05:20

Mathematics, 10.11.2020 05:20



Spanish, 10.11.2020 05:20

Social Studies, 10.11.2020 05:20

Chemistry, 10.11.2020 05:20

English, 10.11.2020 05:20


Mathematics, 10.11.2020 05:20

History, 10.11.2020 05:20


Mathematics, 10.11.2020 05:20


Mathematics, 10.11.2020 05:20





History, 10.11.2020 05:20