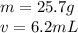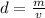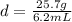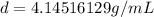Chemistry, 31.08.2020 07:01 homeworkprincess
An irregularly-shaped sample of aluminum (Al) is put on a balance and found to have a mass of 25.7 g. The student decides to use the water-displacement method to find the volume. The initial volume reading is 35.5 mL and, after the Al sample is added, the water level has risen to 41.7 mL. Find the density of the Al sample in g/mL. *

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 16:00
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2

Chemistry, 22.06.2019 20:00
Carbon-14 undergoes radioactive decay in the reaction above. determine the type of radiation emitted in this reaction and describe what is happening to the nucleus during this reaction.
Answers: 2

Chemistry, 23.06.2019 00:00
How many moles of water are created if 3 moles of hydrogen react completely with excess oxygen?
Answers: 3
You know the right answer?
An irregularly-shaped sample of aluminum (Al) is put on a balance and found to have a mass of 25.7 g...
Questions

Mathematics, 18.03.2021 20:10

Mathematics, 18.03.2021 20:10


English, 18.03.2021 20:10

Social Studies, 18.03.2021 20:20


History, 18.03.2021 20:20




Mathematics, 18.03.2021 20:20

Mathematics, 18.03.2021 20:20



Mathematics, 18.03.2021 20:20

Computers and Technology, 18.03.2021 20:20



Chemistry, 18.03.2021 20:20

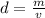
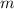 is the mass and
is the mass and  is the volume.
is the volume.